Alien species are giving freshwater ecosystems in Africa and beyond a mauling. But a new project, backed by funding from one of the continent’s premier prizes for young researchers, holds hope.
Yves Vanderhaeghen speaks to Dr Riaan Rifkin about the pioneering gene-sequencing by a team of South African researchers which sheds light on a pathogen which infected a child 2000 years ago.
About 2000 years ago a seven-year-old San boy, feverish and lethargic, lay down and died along the beach at Ballito in KwaZulu-Natal. He was buried just above the high-water line, perhaps by his parents, perhaps by the sediments of wind and time; archaeologists just don’t know what the burial rituals of hunter-gatherers were. His family would not have known why he had died. Now, however, a team of pioneering South African researchers have pieced together the genome of his likely killer: Rickettsia felis, a pathogen which probably hitched a ride on a flea before infecting him.
“Our study was the first in Southern Africa to detect ancient pathogen DNA, the first time we could tell that individual X had a certain pathogen that might had led to his or her death. And this was the first hunter gatherer disease found for the whole of Africa,” says lead researcher on the project, Dr Riaan Rifkin, an anthropologist and prehistoric archaeologist based at the Centre for Microbial Ecology and Genomics at the University of Pretoria. He hopes ultimately to reconstruct how diseases shaped the human condition, both biologically and culturally, from the origins of the species to its exodus from Africa about 60 000 years ago, taking a host of diseases with it.
The Ballito Boy has been a breakthrough in the understanding of not only what diseases afflicted our ancestors, but of the very definition of “human”, according to the findings of the team who sequenced his genome in 2017, and on whose work Rifkin and his team drew. That joint Swedish-South African study found that his DNA timed the split between modern humans and archaic or pre-human groups between 350 000 and 260 000 years ago. The received archaeological wisdom had previously placed the emergence of modern humans at less than 200 000 years ago.
The boy’s remains were uncovered accidentally, according to Dr Gavin Whitelaw, Chief Curator at the KwaZulu-Natal Museum, in a shell midden in the early 1960s. Although the exact location is not recorded, it was about 50 metres from the high-water mark, in the vicinity of where the Ballito Ski-Boat Club is now. The skeleton was handed to Durban Natural Science Museum and then, together with the remains of other individuals, it was moved to the KZN Museum in Pietermaritzburg in the 1990s, where the DNA samples were extracted, and where it still resides.
Rifkin believes that “a lot of the pathogens which still accompany humans across the globe actually spread with humans out of Africa. But no one has ever looked for ancient pathogens from ancient African archaeological contexts”, except for studies of Egyptian mummies which showed that modern diseases such as hepatitis B, diptheria, bubonic plague, smallpox and tuberculosis have ancient origins
But how did it come about that he selected this skeleton to look for pathogen DNA?
“Well, we didn’t actually choose it,” says Dr Rifkin. “We ran through at least four or five other data sets. In the beginning we thought we would be looking for pathogen DNA from sediments in archaeological sites. As humans lived on a shelter floor or a cave floor, they would never clean the floor or vacuum it or wash it or anything. They would live there, they would eat there, they would sleep there. So we thought okay, given the difficulties of working with human remains in South Africa, we would start with sediments, and we ran through quite a few data sets. In some of the sediments we did find pathogens but the DNA fragments are so small and heavily fragmented that you can’ really piece together a proper genome for an ancient pathogen.
“And then we came across a publication by colleagues of ours a few years ago on Ballito Bay, and they were only interested in the human genome. They wanted to see how the human genome fitted the “Out of Africa hypothesis”, and it clarified the fact that our ancestors did evolve in Africa and that we all have an ancestor in Africa. But as you sequence genomes there’s a lot of extra data that comes out. They did shotgun sequencing, which means you sequence every single thing in this child skeleton. And then they extracted the human DNA and focused on that. And we said to them can we have a look at the other DNA; all the meta-DNA, all the leftovers that they were not interested in that are not human.
“They said yes, and that was the start of a long process. Eventually we came up with Rickettsia felis, that infected this child, and we believe it might have played a role in his death. So it wasn’t the first sample or data set that we looked at. We looked at many others and most of them just didn’t work out. The heat in Africa is quite a problem for DNA preservation, and it depends on the soil sediment’s chemistry as well.”
How, then, did this child manage to be in a sufficient state of preservation to retrieve DNA?
“Yes that is interesting. It was buried just above the high water line, where it’s very sandy, not very clay-ey. But maybe the salt content and the heat on the beach is what preserved the skeleton. And maybe that’s what helped preserve the DNA as well. It’s a bit of a mystery. But we actually managed to recover some DNA from the petrous bone, which is located behind the ear, and which no one has ever found any pathogen DNA in before. It’s a very porous type of bone, which becomes very hard as you age, but because this child was about seven years old when he died, maybe the petrous bone was still soft enough to have absorbed a lot of the pathogen DNA given the blood flow through your system.”
From this, then, they reconstructed the Rickettsia felis genome, a bacterium which causes typhus-like rickettsioses. Rifkin notes that it had been viewed as a new or emergent pathogen linked to human illness only in 1994, in Texas. But it has now been shown that it was around at least 2000 years ago “amongst southern African Stone Age hunter-gatherers who did not practice animal husbandry or agriculture, and who did not follow a sedentary lifestyle”. (Rifkin has also shown, in other research, that cancer has also been around for a very long time, and cannot only be linked to contemporary and post-industrial lifestyles).
“Rickettsia is a terrible thing. It relates to typhoid and typhus, which decimated Napoleon’s army in Russia, and it’s definitely something which could decimate a human population, should it start to use mosquitoes for transmission, perhaps. So this is our idea, to use ancient data and try to see how these diseases might affect us, in modern times or in the future, and maybe combat them.”
“The relevance of ancient human diseases,” says Dr Rifkin, “is that 70% of humans in the past would have died from a pathogen before reaching adulthood. Disease was definitely the primary killer. Diseases were the most limiting factor for human survival. So hunter-gatherers would start having children at 13 or 14, and have as many children as possible, and seven out of ten might die, and the survivors would do the same thing again to continue that band, or that family.”
Moreover, “if you look at all the humans pathogens, about 70% of all human diseases are zoonotic, which means they come from animals. We share them with them. Tuberculosis, for example, is shared by seals and buffalo and lion. Rickettsia too is not a species specific pathogen. It’s something that would infect whatever the flea or the tick might bite. He and his colleagues argue that “Our results necessitate further discussion about the susceptibility of humans to, and the population impacts of, zoonotic diseases on human longevity and behaviour in the past”.
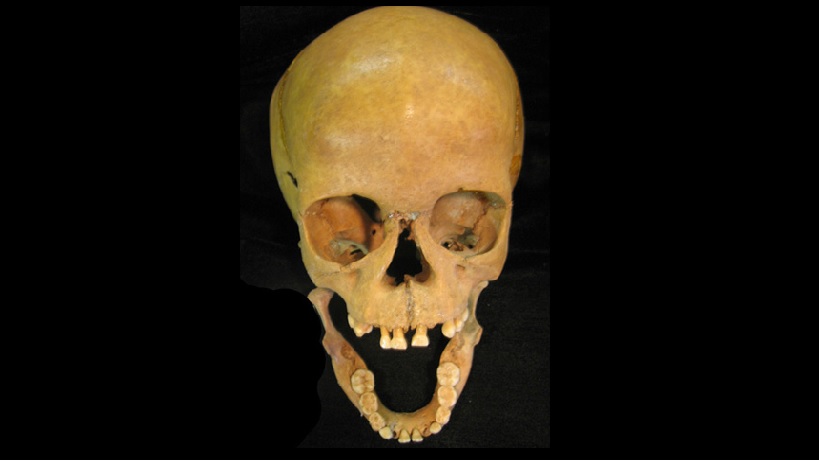
Finding Rickettsia felis in the Ballito Boy has added to the shifts in thinking about how humans lived and evolved. “People believe that most diseases only emerged in the neolithic period (12 000 years ago) when farming and large settlements started. We thought this can’t be true. We’ve always been subject to pathogens. If you lived out here 50 000 years ago, every single pathogen that was around would have infected a human being. And, if you’re ever infected by anything and you survive, that signature of infection would be present in your genome. There would be a certain mutation in your genome which would show, hey, this guy had X disease at some point. And so that’s how we know for example that Covid has been around for about 20 000 years”.
Dr Rifkin notes that although “diseases are not conscious and they can’t think, they don’t really want to kill the hosts because they want to continue spreading. It’s never good for a pathogen to be extremely deadly. It’s good for them to be extremely infectious, because they can spread fast through a human or other population. So Rickettsia felis looks like it was around for long enough for humans to adapt to it, but also for the pathogen to adapt to not killing every single human that it infects.” In the case of the Ballito Boy “he could have been infected with something else too, like malaria. We believe he was infected with other pathogens, we just can’t prove it. With DNA matching at the moment, it’s impossible to say what else he had.”
Felis is not the only Rickettsia around, and the symptoms of infection range from mild to severe and even fatal. In their publication of their results, Rifkin and his colleagues note that, “without antibiotic treatment, murine or ‘endemic’ typhus, caused by R. typhi, exhibits a mortality rate of 4%, and Rocky Mountain spotted fever a mortality rate as high as 30%. Epidemic typhus, caused by R. prowazekii, has a mortality rate which varies from 0.7 to 60% for untreated cases. Mortality rates as high as 66% has been reported for disease due to R. rickettsii occurring prior to 1920, preceding the discovery of antibiotics.” The fatality rate from Rickettsia felis infections can run to 19%, and in Africa it is also the cause of “fevers of unknown origin”.
Where to now for Dr Rifkin?
“At the moment we’re analysing the DNA from the other individuals buried at Ballito. It’s a bit like playing darts. You hope you hit the bull’s eye but it doesn’t always happen. So you need to extract as much DNA as you can, then you need to run the bioinfomatics for all the data sets, and all the DNA, and then you need to see what you get. You could expect a number of different diseases from these individuals, it could be anything. The exciting part of this is that anything is good, because at the moment we have nothing. We don’t know what other diseases these hunter gatherers had.”
In their study of the Ballito Bay Boy, archaeologists Susan Pfeiffer, Lesley Harrington and Marlize Lombard wrote: “It is noteworthy that the bones and teeth of a little boy who lived two millennia ago have provided new insights into the origin and diversification of our species. His story is one of layered improbabilities: That his skeleton was preserved by favorable burial conditions, discovered and donated to a curatorial institution, dated and studied by archaeologists, and chosen for ancient DNA analysis. Based on genomic estimates of large population numbers, his life was one of many thousands that have gone unobserved, yet here we have a small window into the humanity of our ancestors”. And, thanks to Dr Rifkin and his team, we also have a glimpse of the type of pathogen that plagued them.
Sidebar: The Ballito Boy would have been part of a small band of Stone Age hunter-gatherers who roamed along the KwaZulu-Natal coastline, venturing inland from time to time. They would have had the world to themselves, as, 2000 years ago, herding communities with their sheep, goats and cattle had not yet ventured from East Africa to southern Africa, and Iron Age Bantu-language-speaking farming communities only migrated down from West Africa to KwaZulu-Natal in about AD 300–400.
Susan Pfeiffer, Lesley Harrington and Marlize Lombard write that this group were ancestral to the southern Khoe-San, they were lactose intolerant and had no genetic defence against Plasmodium vivax, one of five parasites that cause malaria in humans, and sleeping sickness. They hunted with bows and arrows, used composite poisoned bone arrowheads and short light spears, and fashioned ostrich eggshell beads. They lived off game hunting, fishing, wild plants and shellfish, in particular brown mussels and oysters which would have been plentiful on the Ballito rocks.
Rifkin. R. et al. Rickettsia felis DNA recovered from a child who lived in southern Africa 2000 years ago. Communications Biology (2023)6:240
Schlebusch, C. M. et al. Southern African ancient genomes estimate modern human divergence to 350,000 to 260,000 years ago. Science 358, 652–655 (2017).
Lombard, M. et al. Ancient human DNA: how sequencing the genome of a boy from Ballito Bay changed human history. S Afr. J. Sci. 114, 1–3 (2018).
Pfeiffer, S., Harrington L., Lombard M. The people behind the samples: Biographical features of Past Hunter-Gatherers from KwaZulu-Natal who yielded aDNA. International Journal of Paleopathology 24 (2019) 158-164.
Dr Riaan F. Rifkin is Oppenheimer Endowed Fellow in Molecular Archaeology, Department of Biochemistry, Genetics and Microbiology, Centre for Microbial Ecology and Genomics (CMEG), University of Pretoria, Hatfield, South Africa. He is also Visiting Academic, Department of Anthropology and Geography, Human Origins and Palaeoenvironmental Research Group (HOPE), Oxford Brookes University, Oxford, UK.
Yves Vanderhaeghen writes for Jive Media Africa, science communication partner to OGRC.
- Research tackles the tensions between nature and necessity - October 1, 2025
- Unlocking Africa’s wildlife economies - September 19, 2025
- The Hidden Carbon Power of South Africa’s Burned Grassland - September 5, 2025
Additional News
Alien species are giving freshwater ecosystems in Africa and beyond a mauling. But a new project, backed by funding from one of the continent’s premier prizes for young researchers, holds hope.
Alien species are giving freshwater ecosystems in Africa and beyond a mauling. But a new project, backed by funding from one of the continent’s premier prizes for young researchers, holds hope.





